So you believe you might have COVID-19, and you want to get examined.
Your initial difficulty may be discovering a take a look at, depending on where by you dwell and how ill you at this time are.
A recent study executed with directors from 323 hospitals across the United States identified…
Estimate: “Hospitals reported that severe shortages of screening supplies and prolonged waits for take a look at success constrained hospitals’ skill to keep track of the health of patients and staff….”
Incorporating: “… they were not able to maintain up with screening demands since they lacked total kits and/or the particular person factors … utilized to detect the virus.”
But let us say that you can essentially get a take a look at for COVID-19.
When screening initial started in the U.S. in January, there was only 1 kind of assay that could confirm COVID-19. It relied on a system called RT-PCR, or reverse transcriptase polymerase chain response.
This system isolates and amplifies the viral code of SARS-CoV-2, which is dependable for the ailment we connect with COVID-19.
Many of the exams finished since that day have utilized RT-PCR, which commonly normally takes about an hour to produce a end result.
Now there are many exams out there, using distinct strategies and getting different quantities of time to return success.
So why are there distinct techniques? And how do they all do the job? And how may screening enable us battle the global pandemic?
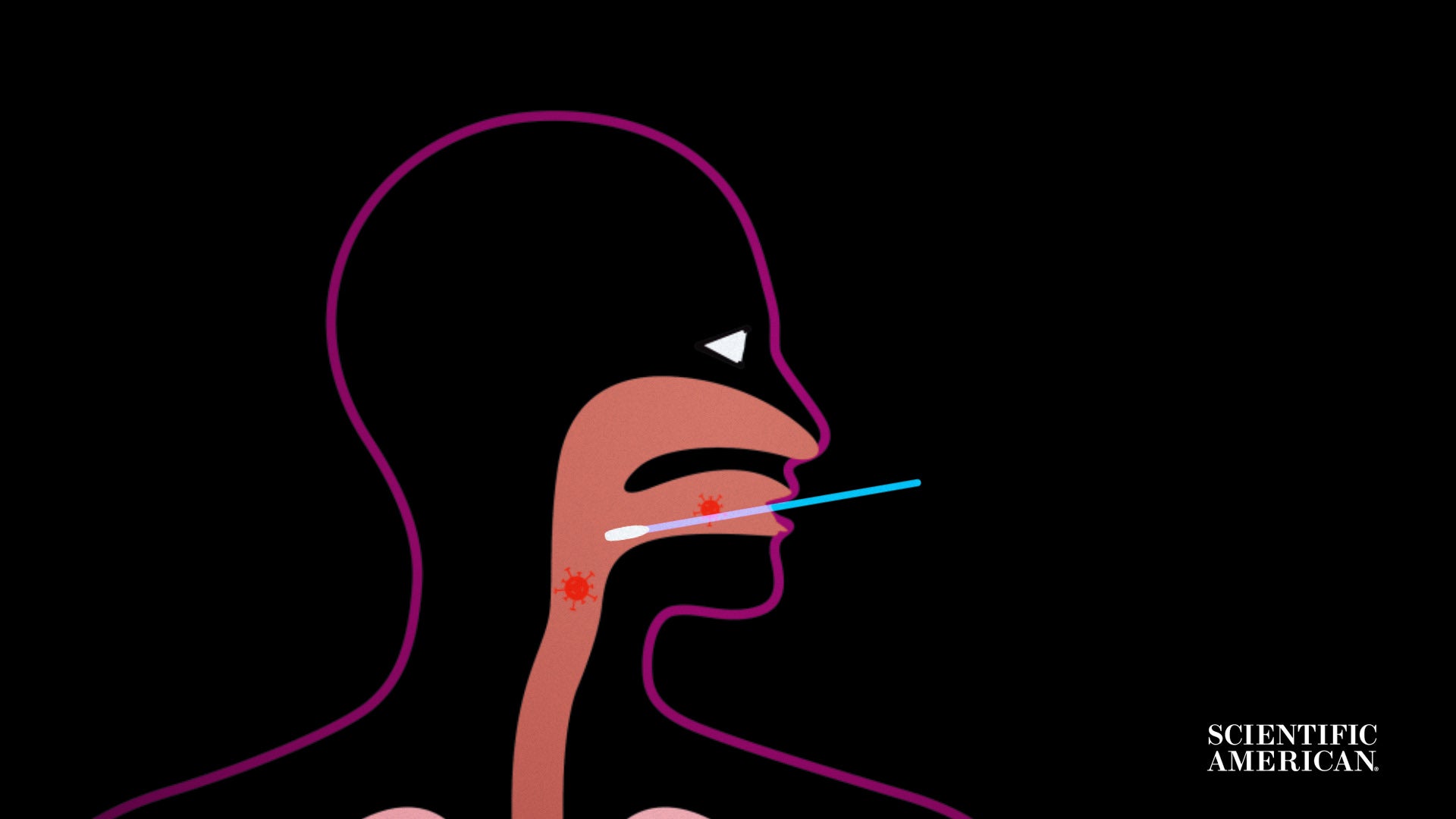
Let us get started with how the virus is gathered from your overall body.
Many exams for energetic COVID-19 infection get started with getting a sample from your higher respiratory tract, where by the virus is regarded to reside.
This indicates pushing a collection swab deep into your nose, throat or nasopharynx, the house that connects the two.
It is time to analyze your sample. Let us get started with the PCR element, or the “polymerase chain response.”
It demands that samples be processed by experienced specialists on specialised machines in screening laboratories.
That indicates patients and the machines that take a look at their samples are possibly far apart.
That transit time can insert hrs or days to finding success again, specially if the screening facility is not close …
… or if there is a backlog in screening.
But when examination commences, the intention is to amplify the viral genetic code. That code is a single strand of RNA.
But the viral RNA should initial be isolated and extracted from your have cells.
Once isolated, the tiny quantities of viral RNA should be amplified to detectable stages.
That’s where by the “RT” or “Reverse Transcriptase” arrives in. It normally takes the single-stranded viral RNA and employs it as a template to produce double-stranded DNA.
That DNA is then copied about and about using the PCR, or “polymerase chain response.”
PCR does this by using cycles of heating and cooling.
It breaks apart the double strand.
Extra chemical substances called “primers” find out certain genes, or portions of that now divided DNA.
RT-PCR exams can goal distinct coronavirus genes that do distinct issues to enable the virus replicate.
Some of individuals genes enable to build the virus’s outer protein envelope. Some make its spiky floor proteins.
The position of this complete system is to exponentially improve the viral DNA with every heating-cooling phase. Right after quite a few cycles of PCR, 1 segment of viral DNA in the sample would flip into hundreds of thousands or billions.
Significant examination machines can also operate several samples at the moment. Even if they just take a handful of hrs to system, quite a few success are returned at 1 time.
But the success of the take a look at should nevertheless be gathered and communicated again to you.
This will cause an additional delay in finding your COVID-19 prognosis.
But although the bulk of ongoing COVID-19 screening will most likely happen this way, new types of exams are starting off to clearly show up.
On February 29th, 2020, the Fda issued an straight away in impact guidance that allows laboratories to submit quick SARS-CoV-2 diagnostic exams to be authorised for use less than an unexpected emergency authorization.
This includes many so-called position-of-care diagnostic exams.
Some of these are just lesser variations of the RT-PCR machines but with prepackaged primers so that any health care employee can operate the sample as long as that person’s facility owns the company’s sample examination machine.
A person of these quick diagnostics employs a new strategy, called isothermal amplification.
Not long ago, President Donald Trump stated 1 business establishing this new type of take a look at.
TRUMP: “On Friday, the Fda authorized a new take a look at formulated by Abbott Labs that delivers lightning-speedy success in as minimal as five minutes. That’s a complete new ballgame….”
The screening machine is tiny, which indicates it can be found in hospitals and doctor’s workplaces. And “isothermal” indicates it detects the virus with no getting to go through the time-consuming heating and cooling cycles that PCR employs. Here’s how it performs.
Less than the hood of this take a look at is a thing called a “nicking enzyme amplification response,” or Near.
It employs primers to goal parts of the SARS-CoV-2 viral code to make a lot more copies with a type of genetic copier-printer template.
Extra enzymes discover the copied parts of viral code and “nick,” or extremely selectively slash, the replicated viral code out of that template like a paper cutter at the close of a printer.
And this printer prints from each finishes, so it spits out two new viral sequences with every new print operation.
Each piece of viral code in your sample can be harnessed by these double-ended genetic copier-printers. This indicates each the printers and the copies from them speedily improve if they detect coronavirus RNA.
Each new viral sequence duplicate also receives a fluorescent beacon—it’s like it leaves the printer with a streak of glowing highlighter on it.
This quick duplicate print of viral sequences is what allows for a constructive ID of COVID-19 in just a handful of minutes.
As of mid April, Usa Right now reported that the business experienced “shipped about 500,000 of the quick exams, which operate on instruments that promote for $4,500 apiece, to all fifty states, Washington D.C., Puerto Rico and the Pacific Islands.”
The final kind of take a look at is a blood take a look at. And it is crucial for discovering out how quite a few of us have been contaminated, and, possibly, who may be able to return to do the job, and when.
It is called an antibody take a look at, immunoassay, or serologic take a look at.
It performs by identifying some of your body’s have defenses from the virus, called antibodies.
These proteins, called immunoglobulins, only exist inside you if you’ve mounted an immune reaction to the coronavirus. These exams usually use a part of the viral code to lookup for these antibodies in your blood.
It is significant to note that these exams are not replacements for the swab-based exams for energetic infections.
The U.S. Fda states as considerably, citing the delay involving onset of infection and antibody build up.
These exams do excel at discovering out who has previously been contaminated.
Knowing how quite a few of us have been contaminated and fought off the virus, at times with mild or no signs, is crucial for long-term disease surveillance.
And if you’ve previously experienced COVID-19 and lived through it, your overall body might know how to battle off reinfection in the future—at minimum for some time period of time.
Just how long your immunity may final is an open issue, while it opens up the chance of individuals who have recovered returning to normal do the job and speak to with other people.
Also, if your immune reaction was sturdy ample at beating again the virus, you might have effective antibodies in your blood.
Those would be useful to other people with weaker immune responses to the virus.
In reality, the Fda not long ago selected so-called “convalescent plasma” as an investigational solution for critically unwell coronavirus patients.
This plasma could also enable in the progress of effective vaccines.
Regardless of what exams you just take for COVID-19 in the coming months, what continues to be clear is that all of these strategies will be vital to beat the coronavirus and close the pandemic.
