The nearer people today are physically to one particular an additional, the greater the chance for trade, of points like suggestions, details, and even infection. Now researchers at MIT and Boston Children’s Medical center have discovered that, even in the microscopic setting in a solitary mobile, physical crowding increases the chance for interactions, in a way that can considerably alter a cell’s health and fitness and improvement.
In a paper revealed now in the journal Mobile Stem Mobile, the researchers have revealed that physically squeezing cells, and crowding their contents, can cause cells to develop and divide more quickly than they ordinarily would.
When squeezing a thing to make it develop could sound counterintuitive, the workforce has an rationalization: Squeezing acts to wring water out of a mobile. With much less water to swim in, proteins and other mobile constituents are packed nearer with each other. And when certain proteins are introduced in near proximity, they can cause mobile signaling and activate genes in the mobile.
In their new examine, the researchers discovered that squeezing intestinal cells induced proteins to cluster together a specific signaling pathway, which can assistance cells manage their stem-mobile condition, an undifferentiated condition in which can rapidly develop and divide into extra specialized cells. Ming Guo, affiliate professor of mechanical engineering at MIT, suggests that if cells can merely be squeezed to advertise their “stemness,” they can then be directed to rapidly create up miniature organs, these as synthetic intestines or colons, which could then be employed as platforms to understand organ perform and check drug candidates for several illnesses, and even as transplants for regenerative medication.
Guo’s co-authors are direct creator Yiwei Li, Jiliang Hu, and Qirong Lin from MIT, and Maorong Chen, Ren Sheng, and Xi He of Boston Children’s Medical center.
Packed in
To examine squeezing’s influence on cells, the researchers combined several mobile sorts in solutions that solidified as rubbery slabs of hydrogel. To squeeze the cells, they put weights on the hydrogel’s area, in the sort of either a quarter or a dime.
“We desired to realize a important amount of money of mobile size alter, and those two weights can compress the mobile by a thing like 10 to 30 p.c of their overall volume,” Guo clarifies.
The workforce employed a confocal microscope to measure in 3D how personal cells’ styles altered as just about every sample was compressed. As they anticipated, the cells shrank with tension. But did squeezing also influence the cell’s contents? To response this, the researchers 1st appeared to see no matter if a cell’s water content material altered. If squeezing acts to wring water out of a mobile, the researchers reasoned that the cells really should be much less hydrated, and stiffer as a end result.
They measured the stiffness of cells just before and immediately after weights have been used, working with optical tweezers, a laser-primarily based system that Guo’s lab has utilized for decades to examine interactions in cells, and discovered that without a doubt, cells stiffened with tension. They also saw that there was much less movement in cells that have been squeezed, suggesting that their contents have been extra packed than standard.
Future, they appeared at no matter if there have been modifications in the interactions among certain proteins in the cells, in response to cells staying squeezed. They focused on a number of proteins that are recognised to cause Wnt/β-catenin signaling, which is concerned in mobile expansion and maintenance of “stemness.”
“In basic, this pathway is recognised to make a mobile extra like a stem mobile,” Guo suggests. “If you alter this pathway’s activity, how cancer progresses and how embryos develop have been revealed to be pretty different. So we assumed we could use this pathway to show how mobile crowding is important.”
A “refreshing” route
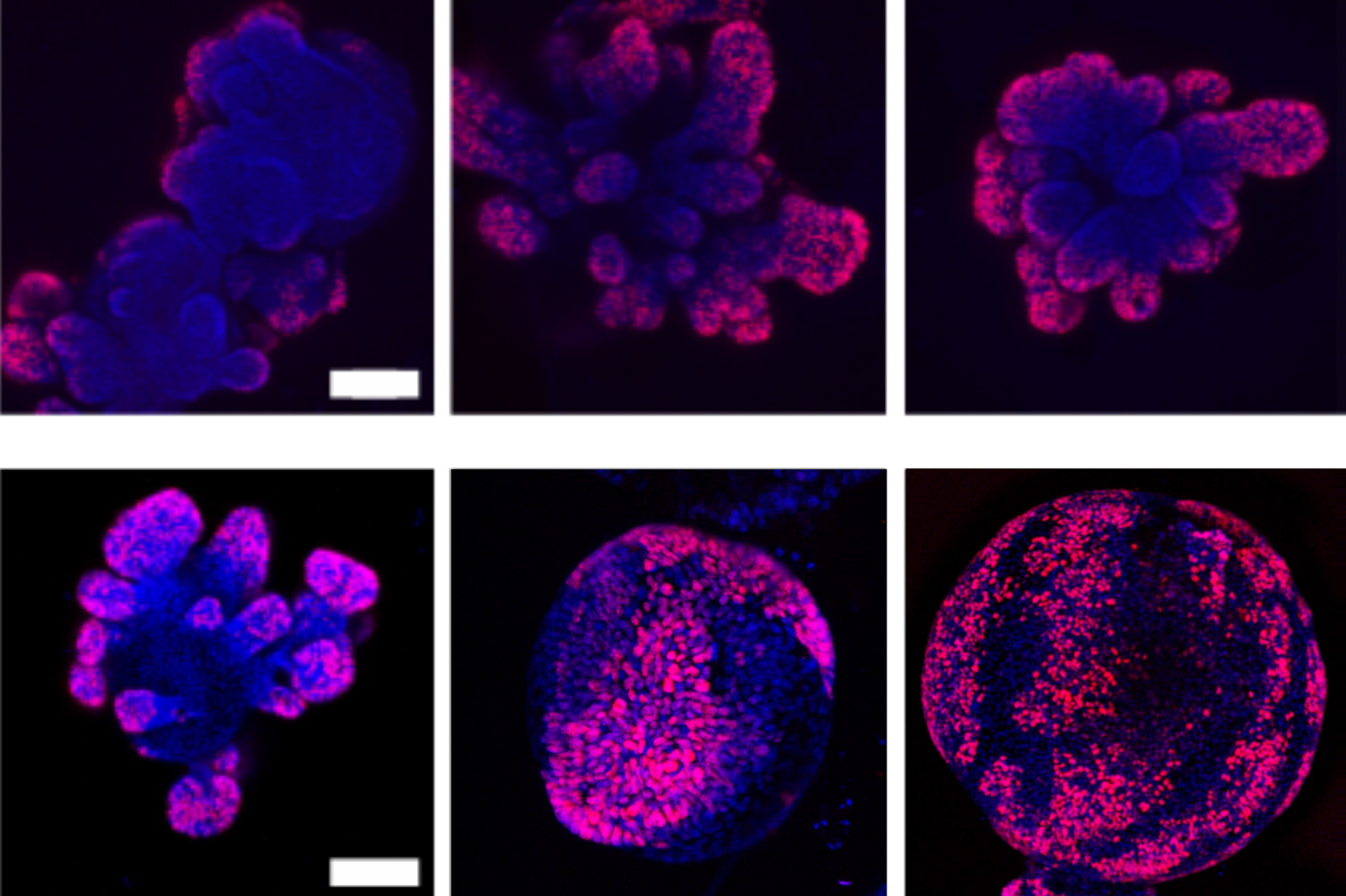
To see no matter if mobile squeezing affects the Wnt pathway, and how rapid a mobile grows, the researchers grew compact organoids — miniature organs, and in this situation, clusters of cells that have been collected from the intestines of mice.
“The Wnt pathway is significantly important in the colon,” Guo suggests, pointing out that the cells that line the human intestine are frequently staying replenished. The Wnt pathway, he suggests, is crucial for sustaining intestinal stem cells, making new cells, and “refreshing” the intestinal lining.
He and his colleagues grew intestinal organoids, just about every measuring about 50 percent a millimeter, in a number of Petri dishes, then “squeezed” the organoids by infusing the dishes with polymers. This influx of polymers increased the osmotic tension bordering just about every organoid and compelled water out of their cells. The workforce observed that as a end result, specific proteins concerned in activating the Wnt pathway have been packed nearer with each other, and have been extra probable to cluster to convert on the pathway and its expansion-regulating genes.
The upshot: Those organoids that have been squeezed essentially grew bigger and extra rapidly, with extra stem cells on their area than those that have been not squeezed.
“The difference was pretty apparent,” Guo suggests. “Whenever you implement tension, the organoids develop even even bigger, with a lot extra stem cells.”
He suggests the effects show how squeezing can influence a organoid’s expansion. The findings also demonstrate that a cell’s behavior can alter based on the amount of money of water that it is made up of.
“This is pretty basic and wide, and the likely impact is profound, that cells can merely tune how much water they have to tune their biological repercussions,” Guo suggests.
Likely forward, he and his colleagues strategy to investigate mobile squeezing as a way to pace up the expansion of synthetic organs that researchers could use to check new, individualized medicine.
“I could just take my have cells and transfect them to make stem cells that can then be made into a lung or intestinal organoid that would mimic my have organs,” Guo suggests. “I could then implement different pressures to make organoids of different size, then attempt different medicine. I envision there would be a lot of options.”
This study is supported, in section, by the Nationwide Most cancers Institute and the Alfred P. Sloan Foundation.
