When Tonya Zerkle, a mother in Cincinnati, Ohio, identified out that Ontario, Canada was vaccinating any small children who switch 12 by the stop of the year, she wasted no time. Immediately after confirming with Canadian officers that her American 11-calendar year-aged was suitable and examining with border authorities on needs to visit, she and her spouse and children headed for the border on a Sunday early morning in late August for one dose of the Pfizer vaccine and one poutine lunch on the way back.
“If I have a way to shield my child, I’m heading to choose it,” says Zerkle, who has apprehensive about her daughter attending a college where by masks are only optional. “But the irony was not missing on me that I was virtually crossing the border to do a thing to safeguard my baby.” She also recognizes that she’s fortunate. “Not a large amount of people might be able to do this,” she adds.
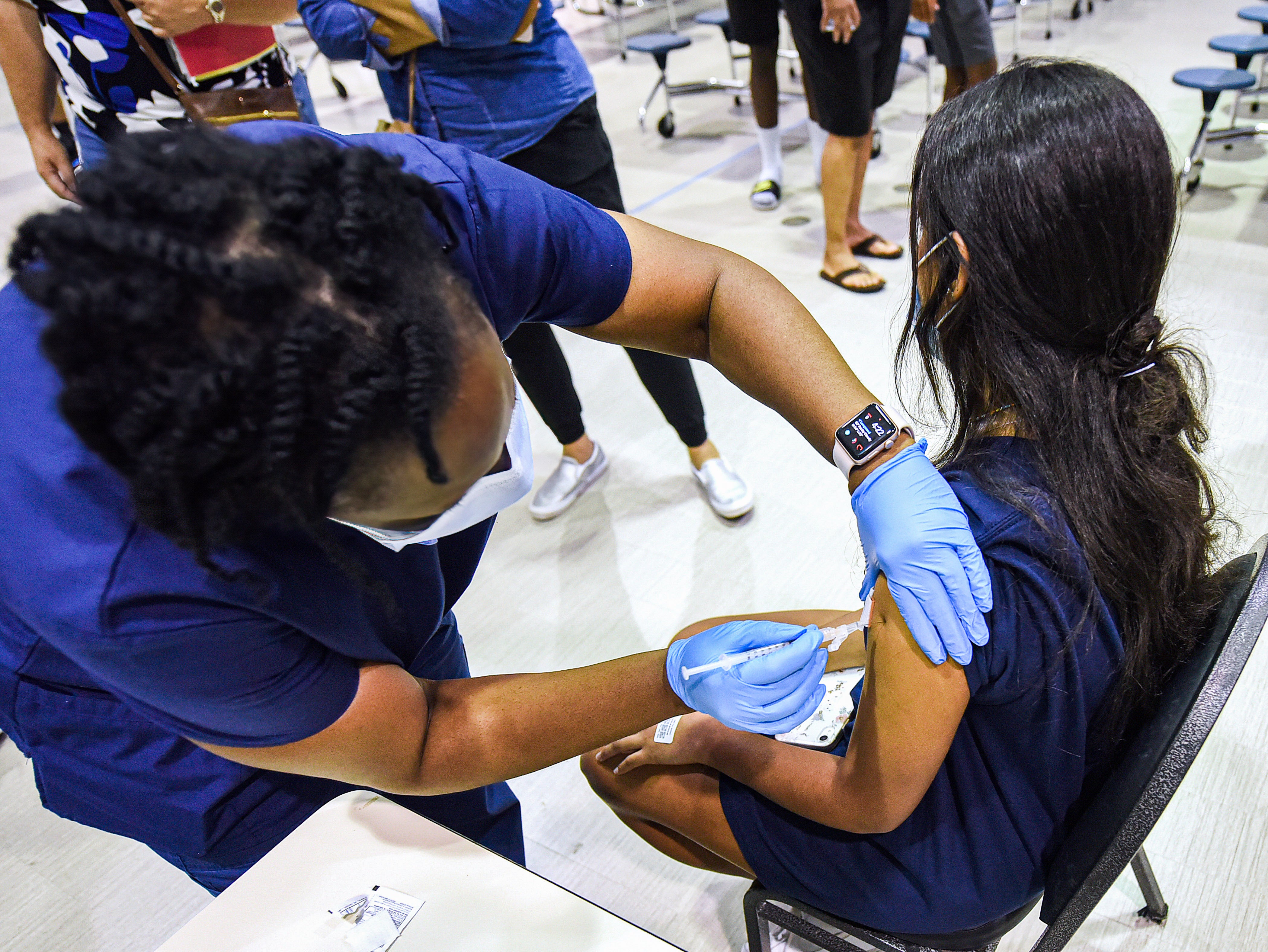
A great deal of dad and mom have by now lied about their preteen’s age to get a COVID vaccine prior to the U.S. Meals and Drug Administration authorizes a single for youngsters under age 12. Thousands and thousands of other mothers and fathers throughout the place carry on to wake up each individual working day with the exact issue: “When will a vaccine for young ones be readily available?”
The response is “really shortly,” states Kawsar Talaat of Johns Hopkins University, a principal investigator for the Pfizer vaccine scientific trials in small children from six months to 11 a long time outdated. In a press release on Monday, Pfizer shared preliminary data from its demo in 2,268 kids aged five by way of 11, showing a “strong immune response” 1 thirty day period just after the second dose that was on par with that noticed in demo individuals aged 16 as a result of 25. The side consequences in the children’s trial were being also related to those observed among younger grown ups, the business documented. Pfizer has not nevertheless unveiled any info on the vaccine’s efficacy in preventing disease in this age group.
“People are working as tricky as they can, as fast as they can to get this details out, and the Fda has promised to assessment it as quickly as feasible. So [the vaccine] will be here before long,” Talaat states.
“Soon” could be as early as the conclude of Oct or maybe early November, Scott Gottlieb, former commissioner of the Food and drug administration and a present Pfizer board member, advised CNBC. The Food and drug administration introduced a assertion on September 10 to handle the mounting concerns and concerns about the timetable for children’s vaccines. “Many of our team at the Fda are parents and grandparents by themselves, and our team shares the exact worries as several in our place about guarding our cherished kinds from COVID-19,” said the statement, which was attributed to Performing Food and drug administration Commissioner Janet Woodcock and Peter Marks, director of the agency’s Middle for Biologics Evaluation and Investigate.
The assertion emphasized the will need for “thorough and strong medical trials” to evaluate the protection and immune response of the COVID-19 vaccine in a populace that is nevertheless rising and establishing. “Conducting clinical trials to ascertain an appropriate vaccine dose in young children needs added get the job done more than that completed in the adult experiments, which include ensuring that the vaccine dosage and formulation strength used is the appropriate a person from the standpoint of safety and building an immune reaction,” it mentioned.
The Food and drug administration laid out the anticipations for a vaccine authorization for small children, such as a prerequisite of a minimal of two months of abide by-up basic safety monitoring in vaccine trials. Immediately after receiving info from the producer, the Fda reported it is “prepared to full its overview as promptly as attainable, probable in a make any difference of weeks rather than months.”
In accordance to Package Longley, senior manager of science media relations at Pfizer, the business expects it will have enough facts by late September to assess its vaccine’s security and efficacy from the medical trials in children aged five to 11. “Pending the data demonstrating protection and immunogenicity, and in agreement with regulators, we could likely post for a U.S. emergency use authorization (EUA) shortly immediately after that,” Longley suggests.
For the nation’s biggest team of pediatricians, having said that, that is not before long more than enough. A 7 days and a fifty percent soon after the Fda requested Pfizer and Moderna to develop the size of their pediatric trials in late July, the American Academy of Pediatrics (AAP) sent a letter captivating to the Fda to think about an early authorization based on details collected so far, offered the skyrocketing pediatric hospitalizations throughout the surge of the novel coronavirus’s extremely contagious Delta variant. “In our watch, the rise of the Delta variant modifications the danger-advantage examination for authorizing vaccines in kids,” the AAP letter read through. It went on to say that “the Food and drug administration ought to strongly look at authorizing these vaccines for youngsters ages 5-11 years based on facts from the initial enrolled cohort.”
At the exact same time, however, the AAP has recognized the danger of vaccinating children prior to the Food and drug administration has issued an EUA for this age group and strongly discouraged medical professionals from administering the Pfizer vaccine—now completely permitted for ages 16 and older—off-label to little ones below age 12. While the group still wants to see the Food and drug administration evaluate the details on 5- to-11-calendar year-olds collected by Pfizer to date as soon as attainable, it does not want pediatricians performing without Fda steering, specifically when all those knowledge are not publicly accessible.
The biggest trouble with off-label vaccination of youngsters beneath age 12 has been the lack of data relating to the appropriate dose, suggests Paul Offit, director of the Vaccine Schooling Center and a professor of pediatric infectious condition at Children’s Healthcare facility of Philadelphia. (Pfizer lastly uncovered on Monday that the pediatric dose for ages 5 to 11 would be 10 micrograms—one 3rd of what grownups receive—but that dosage is not readily available to the general public still.) “If you give a more substantial dose to a smaller child, are you putting them at chance? It’s an mysterious chance,” Offit says.
The Food and drug administration statement likewise noted that the doses for kids more youthful than 12 are diverse than these previously approved for persons age 12 and more mature, even more emphasizing the will need to wait for info from the trials to ensure the vaccine “doesn’t result in unexpected protection troubles different from these that have presently been noticed in adolescents and adults.”
The good news is, no main safety considerations have been noticed so far for the duration of the trials, Talaat states. But she also points out that some rarer security events may not emerge until the vaccine is administered in a larger sized population. This was the situation with rare instances of myocarditis—an irritation of the coronary heart muscle—in a pretty compact share of adolescents who gained either mRNA vaccine.
Evidence has considering the fact that demonstrated that myocarditis is a lot more than 5 instances more likely right after a COVID infection than right after vaccination, but problems about the adverse function have continue to brought about hesitation among the some mom and dad, according to Hina Talib, an affiliate professor of pediatrics at the Albert Einstein School of Medicine and an adolescent medication specialist at the Children’s Hospital at Montefiore in New York Metropolis. But she has listened to from even a lot more dad and mom who are desperate for the vaccine, specially if their children show up at educational facilities with no mask requirements. “It’s so tricky for me to look a mum or dad in the eye who is sitting in front of me, who is emotion worry for the basic safety of their kid, and say that [authorization] is just all over the corner,” Talib says. “It’s just gut-wrenching. That feels as well late, but the very best matter to do is to lay eyes on this information, on the suitable doses, for protection, for have confidence in, for the program to perform as it really should.”
She says mother and father are not improper to fret that their little ones are not safeguarded, so they really should concentration on what they can do to reduce threat. “If we cannot get vaccines into arms of five- to 11-yr-olds, every other grownup in the [school] setting up should be vaccinated,” Talib says. “If not, maybe that is negligence on all of our sections as grownups to safeguard young children.”
Data for children aged two by 5 “could get there soon after” the knowledge for people aged 5 to 11, Pfizer’s Longley suggests. And knowledge for youngsters aged 6 months to two decades ought to be obtainable ahead of the stop of the 12 months, “followed by a opportunity submission to the U.S. Fda for an EUA shortly thereafter” as prolonged as the regulators agree.
“That most likely just isn’t quickly enough for several dad and mom, but we need to have to make positive that all the I’s are dotted and the T’s are crossed,” Talaat states. “It’s actually vital that we get it right for our young children.”
